
Direct measurement for density of seawater is done by maintaining the collected water samples in controlled environmental conditions. When air at 25 ☌ and 1 atm is in equilibrium with water, what is the molar concentration of dissolved CO2 given that air contains 0.037 mole percent CO2 (g) The density of water at 25 ☌ is 0.997 g/mL. Likewise, check the water density of samples having same salinity and different temperatures. At 25 ☌ and 1 atm of CO2 pressure, the solubility of CO2 (9) is 149 mg CO/100 g water. All you need is enter the temperature (both in Celsius and Fahrenheit) and salinity in PPM (parts per million) or mg/L in the suggested fields.Īfter finding the results, you can correlate the density of water at room temperature for different salinity levels. Density of potassium salts in water is plotted as function of wt, mol/kg water and mol/l solution. 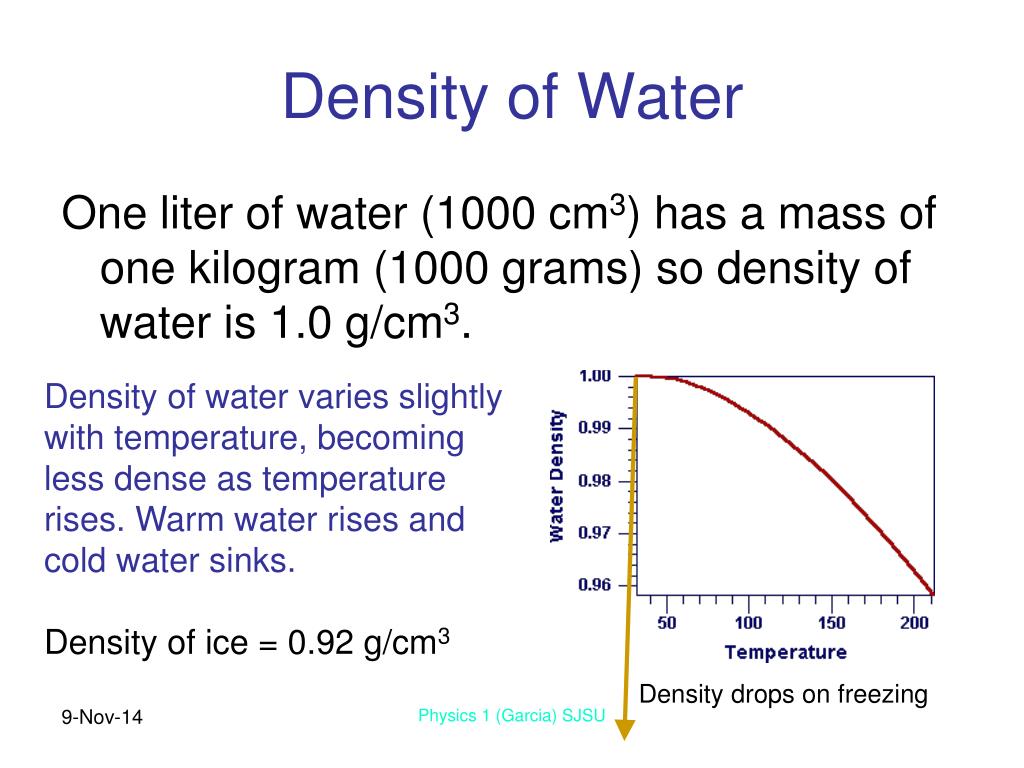
Changes in density of aqueous solutions with changes in concentration at 20☌. If you are interested in calculating the density of seawater at different temperatures, there are online tools that are developed for the same purpose. 25: 921: Water, heavy: 11.6: 1105: Water - pure: 4: 1000: Water - sea: 77 o F (25 o C) 1022: Whale oil: 15: 925: Wheatgerm oil: 25: 926. Thus, water with lower salt content will float over water with higher salt concentration. In short, density of water is directly proportional to salinity. In general, the density of a matter is higher in solid form, as compared to the density of the same matter in liquid form.Īs with salinity, higher the level of salt concentration, the more will be the density of water, even at room temperature. However, as we are discussing the density of water measured in ground level for collected water samples, the pressure attribute is not considered in determining the density. Of course, pressure does play a role in the subject of water denseness. While discussing this topic in detail, water density is found to vary with respect to the level of salinity (or other impurities) and fluctuation of temperature. Below are some selected values of temperature and the saturated vapor pressures required to place the boiling point at those temperatures. If a calorimeter was filled with 104.6 mL of water, what is the mass of water in grams BUY. However, it is to be noted that grams per milliliter (g/mL) and gram per cubic centimeter (g/cm 3) are not SI units. The density of water at 25☌ is 0.998 g/mL. 
The logic is to divide the value of kg/m 3 by 1000 to get pure water density in g/ml. In other words, at the same temperature, the density of water in g/ml (or g/cm 3) is 0.99777. mL Water 5 Make certain the stirrer is On (you should be able to see the shaft rotating). Use the density of the water to determine the mass of water from the volume and record the volume and mass below. The density of water at 25☌ is 0.997 g/mL. At room temperature (i.e., 22° C), the density of water in kg/m 3 is 997.77. The calorimeter has been filled with 100 mL water. With respect to the definition, the density formula is represented as Density (ρ) = Mass (m)/Volume (V).
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
